|
All nuclei contain protons and neutrons, except for the hydrogen which contains protons.the theory is called the Modern Atomic Theory, He came up with the idea that matter is made. However, one of his underlying assumptions was later shown to be incorrect. The neutron mass is almost similar to the proton mass. He came up with his theory of atoms and was said to be accurate. Daltons ideas proved foundational to modern atomic theory.He concluded that the nucleus contains another tiny particle known as a neutron that has no charge.Chadwick discovered the presence of neutrons in the nucleus.Each orbit forms a circle and has a fixed distance from the nucleus.2) All atoms of a given element are identical in mass and properties 3) Compounds. Electrons move around the nucleus in fixed orbits. The Theory of Spectra and Atomic Constitution: Three Essays. Atoms are indivisible and indestructible. 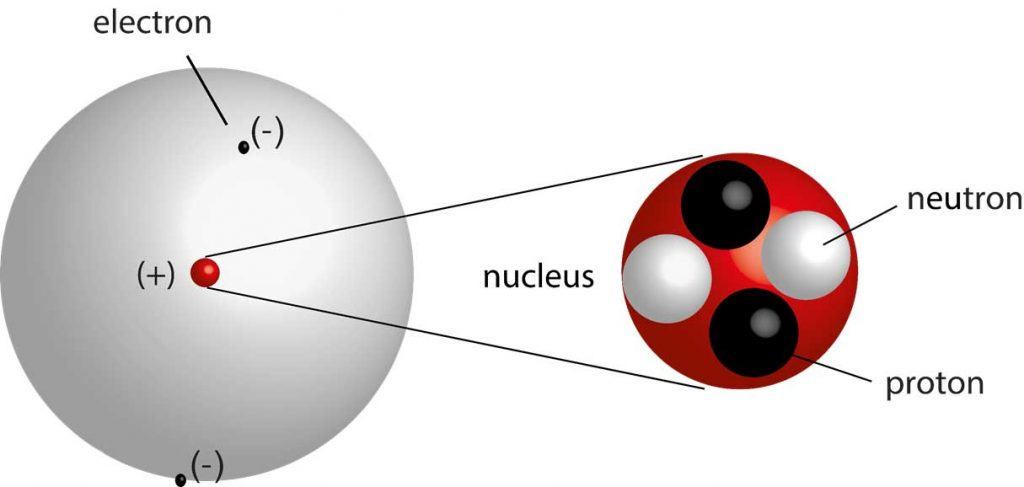
Electrons in an atom of an element are not randomly distributed around the atomic nucleus. 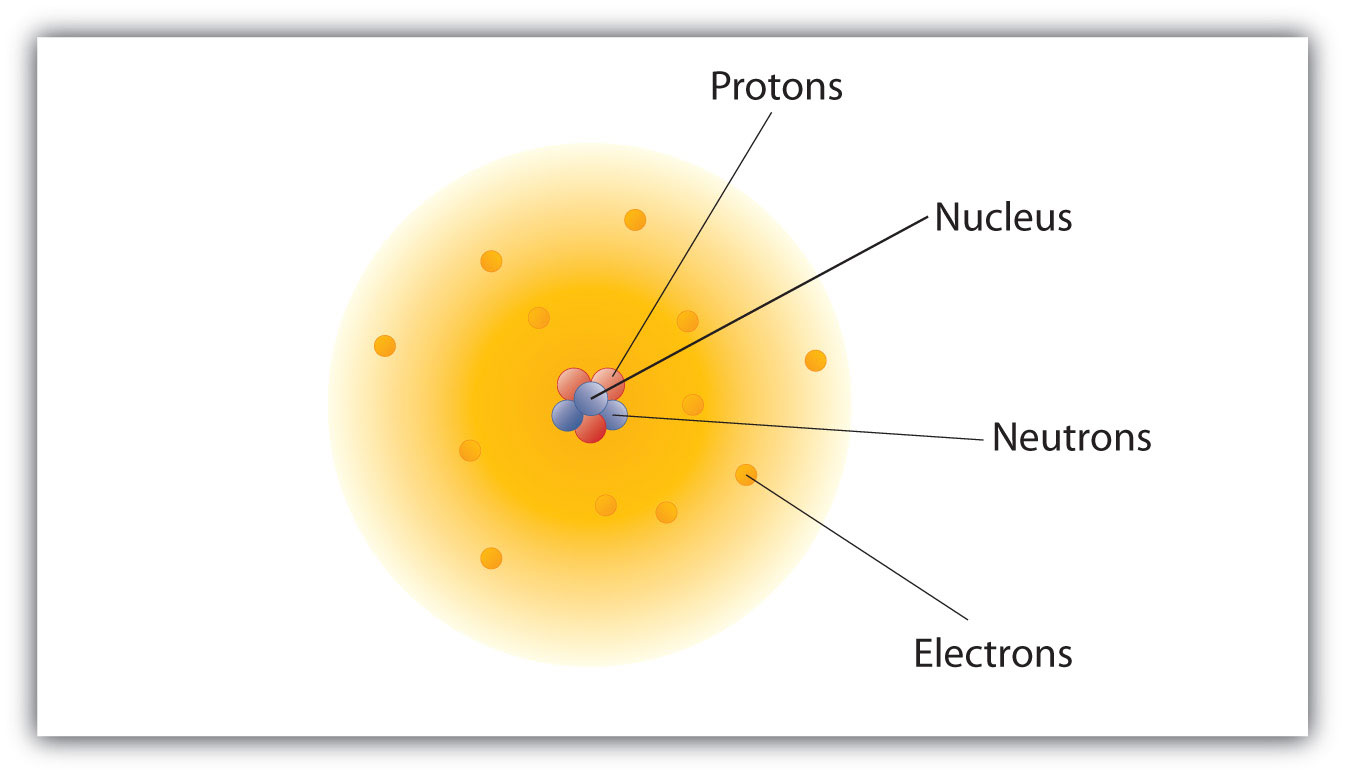
Those alpha particles that had come into close proximity with the nucleus had been strongly deflected whereas the majority had passed at a relatively great distance to it. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
